Implantable ‘living materials’ that deliver drugs on demand could help fight infections
In a “breakthrough,” researchers demonstrate how engineered bacteria held in a jellylike container could help fight infection in mice
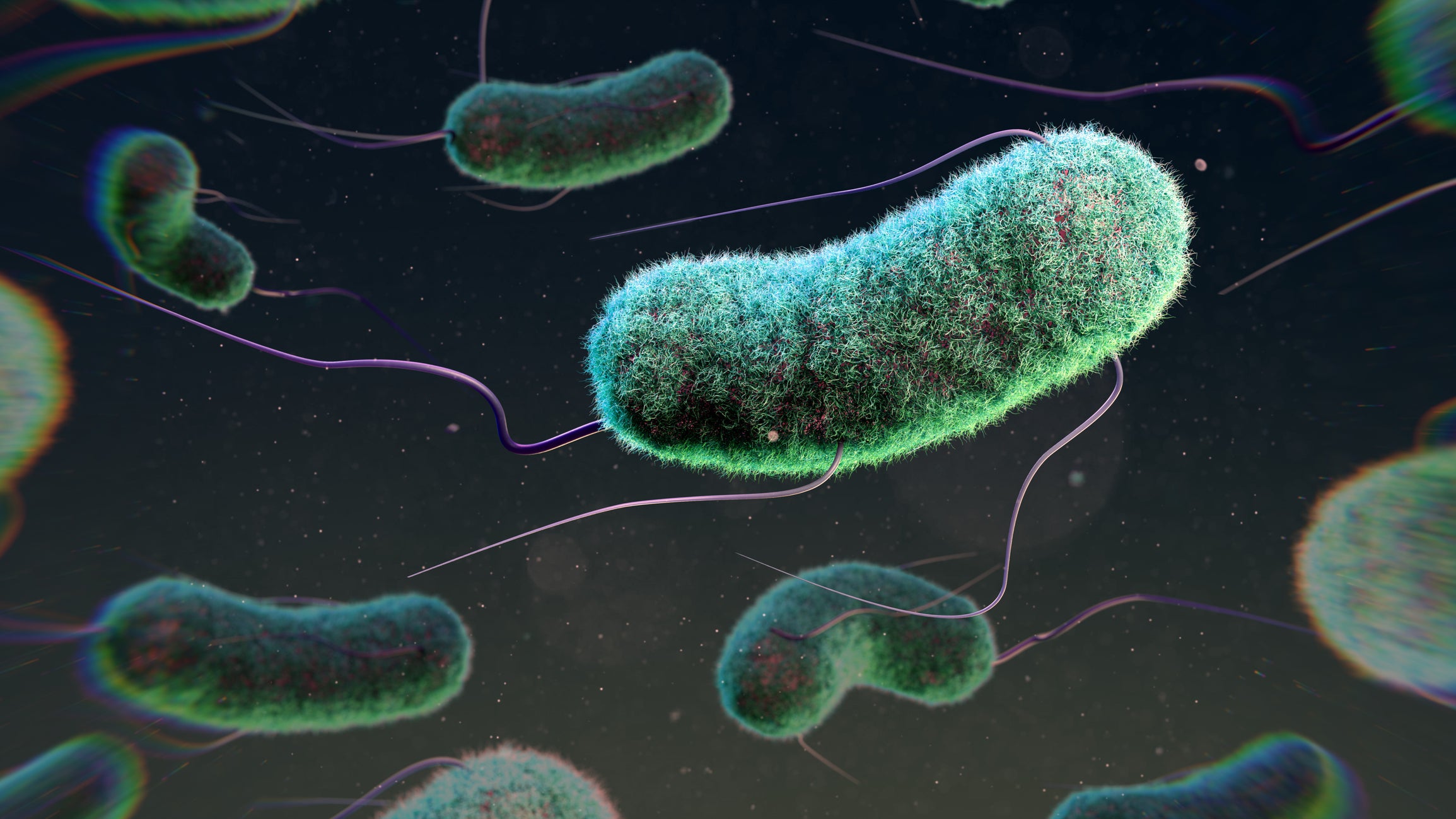
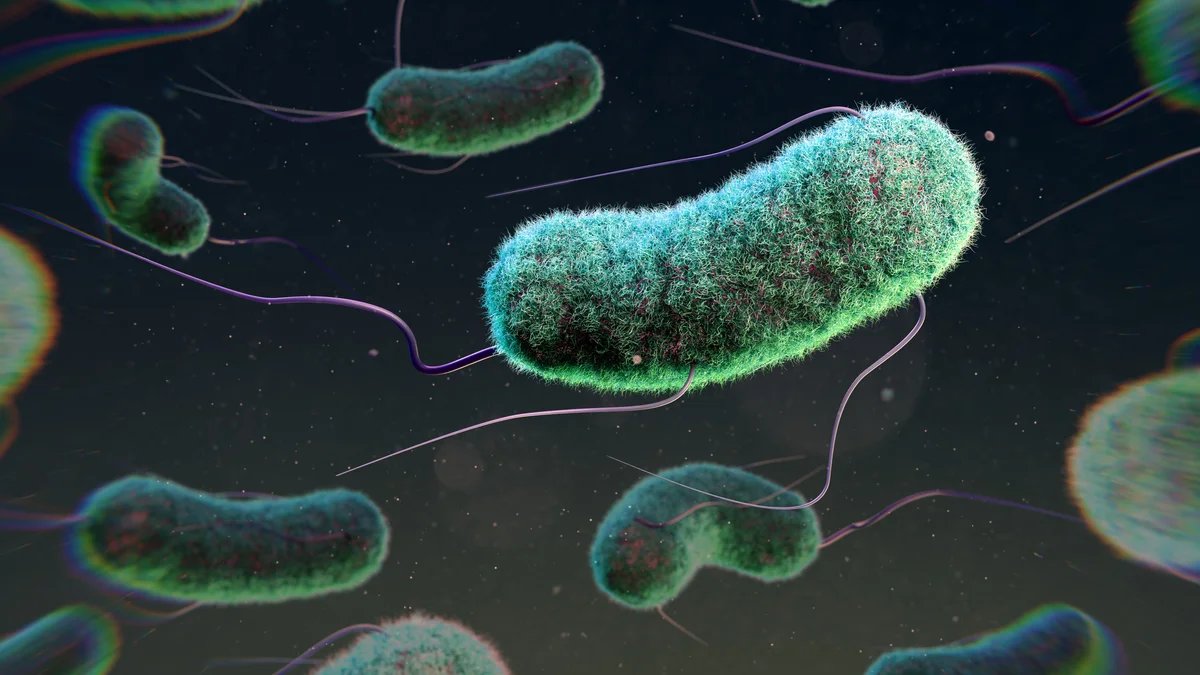
In “living materials,” bacteria such as Escherichia coli could be engineered to sense and fight pathogens.
THOM LEACH/SCIENCE PHOTO LIBRARY/Getty Images
To treat an infection, doctors have to find and diagnose it first. But a new approach involving what researchers are calling “living materials” could fundamentally change that. Designed to sit inside your body and activate when needed to help fight infections at the source, the strategy could dispense with the need for pills, shots and even diagnostics.
In a new study published in Science on Thursday, researchers describe successfully implanting a mouse with a “living material” made with genetically engineered bacteria to treat an infection. The technology, if validated in humans, could be used to help prevent infection in surgeries or as a long-term therapeutic that automatically responds to pathogens.
“Living” medicine isn’t totally new. Scientists already knew that bacteria can be programmed to sense pathogens and self-destruct when they do so, releasing drugs to kill the invader.
On supporting science journalism
If you’re enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
But researchers have been wary about such approaches. “One of the big concerns that everyone has is that we’re injecting live bacteria into our body,” says Tetsuhiro Harimoto, the study’s lead author and a postdoctoral fellow at Harvard University. “We don’t want them to disseminate and cause infections.”
To solve the problem, Harimoto and his colleagues built a “hydrogel,” or jellylike, container. It’s permeable like a mesh yet stiff enough to contain a live and growing population of bacteria without bursting open and strong enough to withstand some wear and tear. The material is similar to the tapioca pearls you might find in boba tea but stiffer, Harimoto says.
To ensure the bacteria wouldn’t escape their hydrogel cage, the researchers ran the hydrogel through a series of stress tests, such as a “fatigue” experiment—in which they stretched the capsule 10,000 times to see if it would break—and let it sit for six months in a “nutrient broth,” Harimoto says.
“Every week or two, we’d basically go in and sample the broth and see if any bacteria [had] come out,” Harimoto says. “At some point, we’re like, ‘Okay, I think this is going to work.’”

Bacteria growth in a hydrogel (scale bar, 50 μm)
The final test involved putting the hydrogel into a mouse. Much like a doctor might put a prosthetic pin in a broken bone to help it heal, the team performed a similar surgery in mice. Some of the rodents received a prosthetic pin treated with a “living” hydrogel that had bacteria designed to detect a pathogen, Pseudomonas aeruginosa, while others didn’t get the treated pin.
To simulate an infection, the researchers also introduced P. aeruginosa to the surgery site. In mice implanted with the “living” hydrogel, the pathogen had a harder time replicating. “We saw a big difference between the one that’s treated with engineered living materials and the one that’s treated with no therapeutics,” Harimoto says.
The results suggest that hydrogels could be used to contain engineered, infection-fighting bacteria for months at a time—a first, says Quanyin Hu, an associate professor in the School of Pharmacy at the University of Wisconsin–Madison, who co-authored a related commentary in Science but was not involved in the study. “The major breakthrough of this study is they can contain these bacteria in the implantation site without the bacteria leaking,” he says.
In theory, Hu says, they technology could also be used to help prevent tumors from coming back, treat inflammation or administer other drugs.
That work is already on the way: in August Harimoto plans to start a lab at Cornell University to test other applications for these programmable “living materials,” including for conditions such as cancer. “Some of the things that we’re excited about [are] to really apply this to a variety of different diseases,” he says.
It’s Time to Stand Up for Science
If you enjoyed this article, I’d like to ask for your support. Scientific American has served as an advocate for science and industry for 180 years, and right now may be the most critical moment in that two-century history.
I’ve been a Scientific American subscriber since I was 12 years old, and it helped shape the way I look at the world. SciAm always educates and delights me, and inspires a sense of awe for our vast, beautiful universe. I hope it does that for you, too.
If you subscribe to Scientific American, you help ensure that our coverage is centered on meaningful research and discovery; that we have the resources to report on the decisions that threaten labs across the U.S.; and that we support both budding and working scientists at a time when the value of science itself too often goes unrecognized.
In return, you get essential news, captivating podcasts, brilliant infographics, can’t-miss newsletters, must-watch videos, challenging games, and the science world’s best writing and reporting. You can even gift someone a subscription.
There has never been a more important time for us to stand up and show why science matters. I hope you’ll support us in that mission.